| Chromate Removal |
| ProSelect HexChrome is a unique weak base anion exchanger with a secondary hybrid capture mechanism for chromate. Under neutral to slightly acidic conditions, chromate is first exchanged into the resin, then reduced to trivalent chrome which covalently bonds to the resin backbone. Throughput capacity is many times greater than that provided by the ion exchange groups alone, allowing very high loading and infrequent change-outs. Because the hexavalent chromate reduction step is both time and pH dependent, it is the rate controlling step. Operation at pH greater than 6 requires low flow rates, rest periods, or periodic soak steps at lower pH to allow the reduction step to catch up. Capacities in excess of 5 lbs of chrome (as Cr) per cubic foot of media are routinely achieved with ProSelect HexChrome when operated at optimum pH and flow conditions. ProSelect HexChrome is not affected by common ions such as nitrate, sulfate, or chloride but can be damaged or fouled by high levels of suspended solids, iron, manganese, chlorine, etc. |
| |
| |
 |
| |
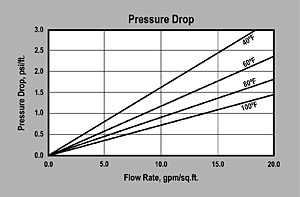
| PRESSURE DROP — The graph above shows the expected pressure loss per foot of bed depth as a function of flow rate at various temperatures. |
| |
| |
 |
| |
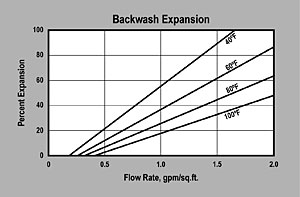
| BACKWASH — The graph above shows the expansion characteristics as a function of flow rate at various temperatures. |
| |
| |
 |
| |
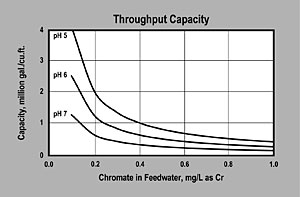
| CAPACITY — The graph above is based on waters with TDS less than 500 ppm and is for chromate alone, exclusive of other anions. Capacity shown is for the working bed in a worker polisher configuration. No engineering downgrade has been applied. |
|
|
| |
Features: |
|
| |
• |
Complies with US FDA regulations (paragraph 21 CFR173.25) for potable water applications |
|
| |
• |
High capacity media designed for one-time use |
|
| |
• |
Large granules provide good physical strength and minimal fines provide low pressure loss |
|
| |
• |
Removes chromium from water using weak base anion exchange resin at near-neutral pH. |
|
| |
• |
Certified to NSF/ANSI Standard 61 |
|
|
| |
| PHYSICAL PROPERTIES |
| Part Number |
ER20011 |
| Polymer Structure |
Epoxy polyamine |
| Polymer Type |
Gel |
| Functional Group |
Mixed amines |
| Physical Form |
Granules |
| Ionic Form, as shipped |
Acid chloride |
| Total Capacity |
2.1 meq/mL min. |
| Water Retention |
52 to 58% |
| Approx. Shipping Weight |
40 lbs./cu.ft. |
| Screen Size (U.S. Mesh) |
12 to 40 |
| Max. Fines Content (<50 mesh) |
1% |
| Uniformity Coefficient |
2 approx. |
| Resin Color |
Amber to yellow |
|
| |
| SUGGESTED OPERATING CONDITIONS |
| Max. Continuous Temperature |
100°F (38°C) |
| Minimum Bed Depth |
24 inches |
| Backwash Expansion |
25 to 50% |
| Max. Pressure Loss |
20 psi |
| Operating pH Range |
4 to 7 SU |
| Service Flow Rate |
1 to 4 gpm/cu.ft. |
|
| |
|
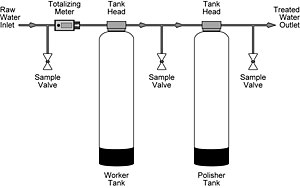
SUGGESTED SYSTEM CONFIGURATION |
 |
| |
| |
|
* CAUTION: DO NOT MIX ION EXCHANGE RESINS WITH STRONG OXIDIZING AGENTS. Nitric acid and other strong oxidizing agents can cause explosive reactions when mixed with organic materials such as ion exchange resins.
|
| |
ADDITIONAL INFORMATION:
SWT Ion Exchange Resin Guide
|
| |
| |
|
This information has been gathered from standard materials and or test data that is believed to be accurate and reliable. Nothing herein shall be determined to be a warranty or representation expressed or implied with respect to the use of such information or the use of the goods described for any particular purpose alone or in combination with other goods or processes, or that their use does not conflict with existing patent rights. No license is granted to practice any patented invention. It is solely for your consideration, investigation and verification.
|
| |
| |
| ProSelect™ is a trademark of Safe Water Technologies, Inc. |
|




